Last month a decision was made by the EFSA (European Food Standards Agency) to put a hold on approving CBD as a Novel Food. It was cited that there was not enough data yet on the safety of CBD, and gaps in the data about potential hazards related to CBD.
The European Commission considers CBD to be a Novel Food product but requested feedback from the EFSA to agree on its approval. However, the EFSA revealed that further evaluations needed to be completed to ensure safety for human consumption.
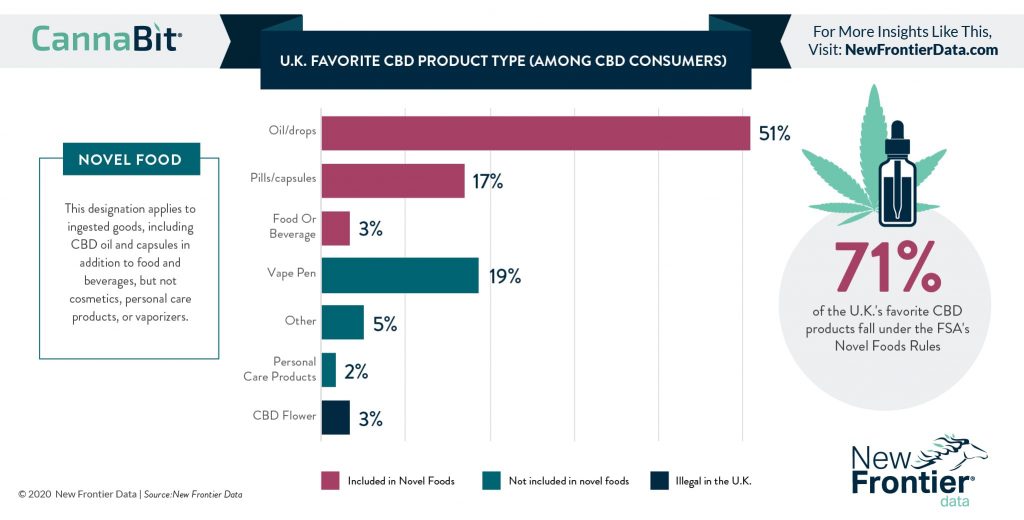
What is a Novel Food and why is it important?
A Novel Food is defined as a food product that has not been consumed to a significant degree by humans before 15th May 1997, when the regulation first came into force. These pertain to newly developed, innovative food produced using new technologies and production processes. To be listed as a Novel Food, the items must be:
- Safe for consumers
- Properly labelled, so as not to mislead consumers
- If Novel Food is intended to replace another food, it must not differ in a way that the consumption of the Novel Food would be nutritionally disadvantageous for the consumer.
Is this classification important for CBD? In short, yes. Having some sort of regulation that can ensure CBD products are safe and are not misleading, is important for customers and brand reputation.
A hold on approval isn’t necessarily a bad thing
There are some who might think this hold on approval is a bad thing for CBD but actually, it can be seen as a positive. The EFSA has noted that there are gaps in the data, but they have not outright suggested CBD isn’t safe for human consumption. The move to put it on hold in the EU is possibly a sign that they are considering following in the UK’s footsteps and requesting more data to be established. In the UK, we have already made the move to list CBD as Novel Food products and despite the EFSA’s decisions, the UK won’t be pulling CBD products from shelves, as the UK FSA have already taken steps to begin establishing regulations.
In the UK many CBD products are listed on the FSA’s website, those of which are legally allowed to be sold. They are still awaiting further assessment but many of them are allowed to be sold on shelves in the meanwhile, as there have been no immediate concerns raised.
Regulations for CBD is not something manufacturers should fear, in fact establishing regulations for CBD can only help to weed out illicit CBD products. Those that don’t contain the amount of CBD they claim or use sub-par, potentially harmful CBD (or none at all!), can easily deter consumers from trying CBD as the results from these products will only ever be lesser. Ensuring that consumers only have access to good quality products is how we can continue to grow the CBD market.
Why you should think about joining the CBD market
Even with the question of regulatory compliance hanging in the air, now is a great time to get ahead and prep your next CBD launch. CBD continues to be popular in the UK and overseas, and regulations only help to ensure that quality products are those that hit the market. Here at Xyfil we continue to produce high-quality CBD products, products that are listed on the FSA’s website, which means quality and safety for consumers.
Let us help get you to market with our experience in product development, manufacture and production. Not only this but we can help get your products onto shelves with our compliance team and understanding of market trends.